Reaching the market isn’t the end of the road for your molecule. Phase IV solutions ensure that your molecule’s potential is fully realized.
Maximize the value of your portfolio and your long time investments
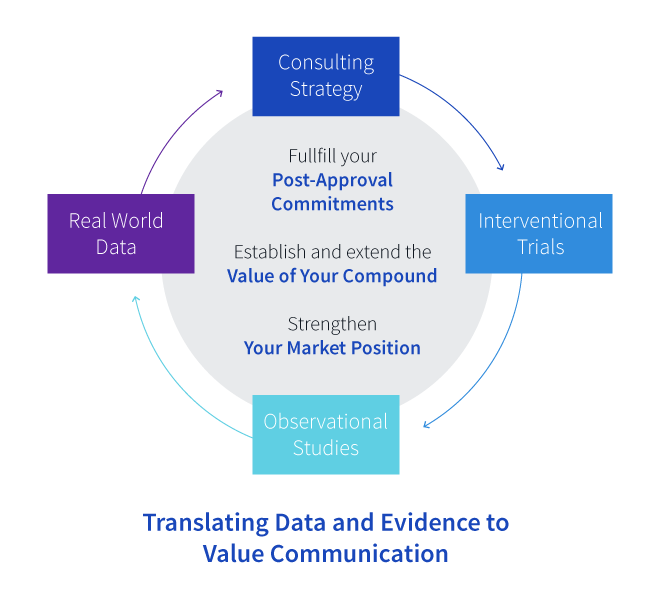
We have over 30 years of experience world-wide in the late phase industry. We have deep institutional expertise across all therapeutic areas, established investigator relations and broad real world insights. This unique combination can help you define strategies and solutions to efficiently generate and communicate evidence that enables product value demonstration for various stakeholders and to:
Timely fulfill your post approval commitments.
Successfully collect Real World Evidence (RWE) and extend the value of your compound.
Strengthen your market position and support uptake.
Fill the gap between registration requirements and strategic late phase study designs.
Engage with patients in meaningful ways and through Patient Reported Outcomes (PROs).
We can maximize the value of your compound through an appropriate design to strategically gather and disseminate real world data
Our medical affairs and post-marketing solutions team will partner with you to deliver customized solutions to meet your design, peri- and post-approval needs.
Preparing for and successfully reaching the market is only the beginning of your compound’s journey. We combine unique perspectives and precision delivery to help you proactively plan your asset’s path to success from the start.