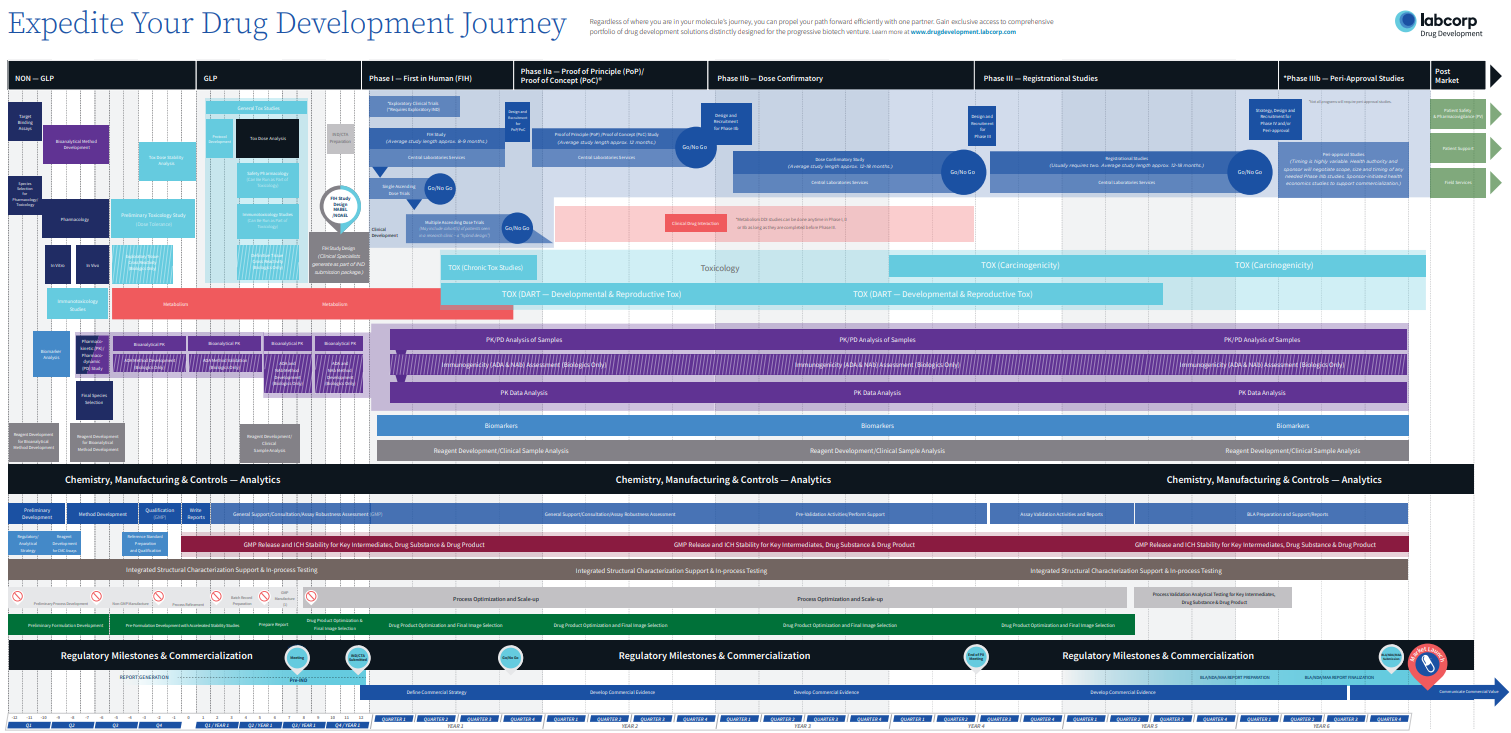
How far you go is up to you
Whether you plan to complete an IND/CTA-enabling program or you need to gain the clinical insight that a first-in-human (FIH) or proof-of-concept (PoC) study can provide, you can enjoy the journey with a dedicated team and a singular, cohesive strategy that transitions seamlessly between nonclinical and clinical development.
Which candidate is best?
Get integrated solutions to rapidly identify and develop your best lead candidate. From early characterization and formulation on development batches, to non-GLP screening for early identification of pharmacology, or toxicity-related issues—rest assured, you’ll move your best candidates forward.
IND/CTA-enabling nonclinical assessment
Take advantage of the vast knowledge of an expert team who manages drug development programs to support hundreds of regulatory submissions each year. With Early Phase Development Solutions, you seamlessly integrate the complete array of nonclinical services, including lead optimization, safety pharmacology, toxicology, pathology, bioanalytical, drug metabolism and pharmacokinetics, to assure successful design and conduct of your program—all the way through IND/CTA submission and into first in human clinical studies.
First-in-Human (FIH) Studies
With Early Phase Development Solutions, you benefit from the retained knowledge from nonclinical study results to move your compound across drug development phases more effectively. The focus will be on two critical aspects of your FIH studies: scientific integrity and human subject safety. As early research continues to demand more complex studies requiring special populations, multiple endpoints and adaptive protocol designs, you’ll gain the advantage through 35+ years of insights and industry-leading human AME expertise.
Proof of Concept (PoC)
Waiting until you have a complete data package before designing your Proof of Concept (PoC) study can waste valuable time. Instead, you’ll enjoy innovative approaches to these shorter, scientifically demanding studies by parallel processing study feasibility and site assessments, incorporating relevant biomarkers and leveraging adaptive trial designs. Increase your clinical ROI by applying the right level of medical, scientific and therapeutic expertise resources and patient stratification strategies to your program.
A Programmatic Approach – Why it Matters
How is it possible to shave up to 30% on your program? It's about streamlining your journey, taking out whitespaces, and rethinking risk-management. Read more in the “Estimate the Impact of Time Savings on Your Drug Development Program, Asset Value and Financial Company Performance” whitepaper. What’s more, we can sit down and do an economic valuator session – to estimate the likely time savings for your specific program.